Featured image: The Golden Toad was last recorded in 1989. Source: U.S. Fish and Wildlife Service (obtained from Wikimedia Commons)
The “Assessing Risk to Endangered and Threatened Species from Pesticides” National Academy Sciences (NAS) report was released in 2013. The report can be read or downloaded here for free: https://www.nap.edu/catalog/18344/assessing-risks-to-endangered-and-threatened-species-from-pesticides.
Reason for committee formation and scope of report
The U.S. Endangered Species Act requires federal agencies to consult with the Fish and Wildlife Service (FWS) or National Marine Fisheries Service (NMFS) when their actions might affect threatened or endangered species (hereby called listed species). The Environmental Protection Agency (EPA) thus consults with the FWS/NMFS (hereby called Services) when registering or reregistering pesticides that might affect listed species.
However, the EPA and Services determine risks to listed species differently and quite independently, resulting in disagreements between the two agencies. Hence, they asked the National Research Council (NRC) to examine the scientific and technical issues related to determining risks to listed species from registered pesticides. The NRC convened the Committee on Ecological Risk Assessment which prepared this report. The committee comprised of people in academia, consulting firms, and a state regulatory agency.
The two laws and why they clash with one another
The Endangered Species Act (ESA): It states that all federal agencies must ensure their actions do not harm listed species. If they believe their actions may harm listed species, they should consult with the Services. An informal consultation is adequate if their actions are not likely to adversely affect listed species or their habitat. However, if their actions are likely to adversely affect listed species or their habitat, a formal consultation is required. At the end of it, the Services shall issue a biological opinion (BiOp) based on the best available data. In the case of pesticides, the BiOp will state whether the EPA has ensured its registration of the pesticide 1) has not killed or injured listed species, either directly or through the destruction or modification of their habitat and 2) has not jeopardized the continued existence of listed species or resulted in the destruction or adverse modification of their habitat. If the Services finds the EPA guilty on these counts, they will develop measures and alternatives that the EPA can adopt to minimize harm or reduce the likelihood of harm to listed species. If the EPA decides not to adopt these measures and alternatives, they could be liable to penalties.
Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA): EPA registers or reregisters pesticides (pesticides have to be reregistered every 15 years) under this act which states that there must be no unreasonable adverse effects to humans or the environment when accounting for the economic, social, and environmental costs (risks) and benefits of pesticide use. The pesticide companies provide data for studies that EPA has determined are necessary to assess pesticide risks. These data requirements are for ‘general pesticide use’, i.e., the companies do not need to provide information on whether the pesticide may harm species at certain locations or times of use. EPA then evaluates the data and, if the pesticide is found to comply with FIFRA standards, it issues a registration. No permit or approvals are needed thereafter (exceptions are restricted use pesticides) but the EPA can require additional data, including data on potential risk to listed species, from companies after pesticide registration. If companies fail to provide the data, EPA can suspend the pesticide’s registration if they believe the continued use of the pesticide will cause unreasonable hazard to listed species.
How the two laws conflict (summarized):
| Endangered Species Act | Federal Insecticide, Fungicide, and Rodenticide Act |
| Administered by the Fish and Wildlife Service and the National Marine Fisheries Service | Administered by the Environmental Protection Agency |
| “Federal agencies must ensure their actions do not harm listed species or their habitat” | “Pesticides must not cause unreasonable adverse effects to humans or the environment” |
| Provides specific protections for listed species | No specific protections for listed species |
| Does not consider economic benefits of federal actions (i.e., use of pesticides) | Considers economic benefits of using pesticides |
| Requires (pesticide) effects be analyzed on individual members of listed species and their habitat | Pesticide registration and cancellation decisions are made on nationwide basis |
| Decisions based on best available data (additional data cannot be requested) | Decisions based on stipulated data that are provided prior to pesticide registration (additional data can be requested) |
Committee recommendations (or how to solve this conundrum)
The committee outlined three steps that could be followed to assess pesticide risks to listed species. This would require greater collaboration between the agencies and would streamline and harmonize the process. Each of the three steps would be coordinated with ecological risk assessment process (i.e., problem formulation, exposure analysis, effects analysis, and risk characterization). The figure (taken from the report) and steps below explain this more effectively.
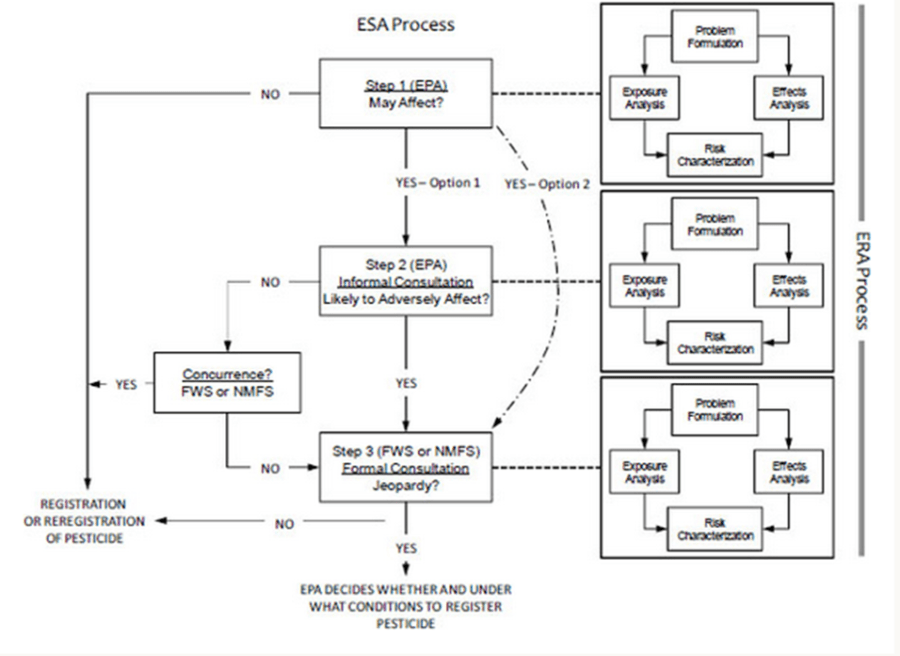
Step 1: EPA would consider whether any listed species might be harmed by the pesticide by checking if the areas proposed for pesticide application coincide with known (or suspected) habitats of listed species.
Step 2: EPA would determine if the proposed use of pesticide would result in environmental concentrations that have the potential to affect listed species and/or their habitat. This would be a generic (not site-specific) assessment and effects on individual members of the listed species would be analyzed. If the predicted concentrations of the pesticide could adversely affect individuals in the listed species population, EPA would consult with the appropriate Service (FWS/NMFS).
Step 3: The Service would determine if local (site-specific) concentrations of the pesticide would affect the listed species (directly or indirectly) in a manner that would change the ability of their population to persist or recover.
The committee felt that increased all-around cooperation between the agencies would effectively combine EPA’s expertise in pesticides with the Services’ expertise in listed species. They also provided other recommendations to harmonize the risk assessment process between the agencies. Most of them are listed below:
a) Since the EPA and Services use different sources of data to make decisions, the committee suggested a broad data search be conducted at the beginning of any assessment. The data must be screened for relevance and then reviewed for quality. Since stakeholders (like pesticide companies) can provide valuable and relevant data, they must be involved in the assessment.
b) Effect of the pesticide needs to be determined for a range of concentrations including all values that a population might encounter (EPA currently determines the effects at certain concentrations like the LC50).
c) Pesticides contain the active ingredient (the main chemical that harms the pest) plus inert ingredients (other chemicals that increase the effectiveness of the active ingredient). EPA mostly assesses the effect of the active ingredient only. The committee suggested the EPA also consider the effect of the inert ingredients if there are data to show it causes an adverse response.
d) The EPA and the Services define sublethal effects differently. The committee suggested that sublethal effects be defined as any effects on survival and reproduction. If indirect effects (e.g., loss of food source due to pesticide use) on the species can be quantified, they should also be included in the assessment.
e) Listed species could be exposed to multiple chemicals and stressors (like climate change). The committee advised that the combined risk of all chemicals be determined and that the cumulative effects of all stressors on listed species be predicted through the use of population models.
f) When toxicity data for listed species are not available or cannot be easily obtained, a surrogate species could be used to assess their risk. Also, instead of using a single surrogate species, multiple surrogate species could be used to obtain a range of sensitivities to the pesticide and estimate risk to the listed species.
g) There always exists uncertainty when it comes to assessing pesticide risks. The committee recommended a probabilistic approach where uncertainty in estimating exposure and effects are combined to yield risk as a probability with associated uncertainty.
h) To ensure the risk assessment has been done in an objective and transparent manner, the committee suggested incorporation of external peer reviews.


1 Comment