The “Preparing for Future Products of Biotechnology” National Academy Sciences (NAS) report was released in 2017. The report can be read or downloaded here for free: https://www.nap.edu/catalog/24605/preparing-for-future-products-of-biotechnology. All images used in this post were obtained from the report.
Reason for committee formation and scope of report
Given the profusion of biotechnology products in recent years, in 2015 the Office of Science and Technology Policy tasked EPA, FDA, and USDA to commission an external, independent analysis of the future landscape of biotech products. The analysis should identify (1) potential new risks and frameworks for risk assessment and (2) areas in which the risks or lack of risks relating to the products of biotechnology are well understood.
The committee largely comprised of people in academia but representatives from other organizations (national lab, company, NGO) with the necessary expertise to carry out the task were also selected. My major advisor, Dr Steven Bradbury, was one of the committee members.
Future biotech products
The committee first grouped future products into three major classes: open-release products (deliberate release into environment), contained products (not released into environment), and platforms (tools used to create other biotech products). Thus, biotech products were broadly defined and can include recombinant and synthetic nucleic acid molecules and proteins, microbial pesticides, intergeneric microorganisms (formed by combining combining genetic materials from organisms in different genera), and genetically engineered (GE) insects, plants, and animals.
Regulating future biotech products
The committee explored if current laws allow for adequate regulation of future biotech products. Federal agencies need to perform two kinds of regulatory functions to ensure human and environmental safety:
a) Consumer and occupational safety to people who expose themselves to biotech products (injuries due to product).
b) Ecological (non-human) risks and risks to humans exposed to biotech products regardless of choice (due to contamination of environment and introduction of pernicious species).
The U.S. typically regulates based on human, ecological, and economic conceptions of risk, but new products can pose other possible harms like loss of employment, changes in societal structures, cultural affronts, loss of biodiversity and its intrinsic values, and could increase inequities.
The major regulatory agencies are EPA, FDA, and USDA. EPA can regulate biotech products under FIFRA, FFDCA and TSCA and FDA can regulate under FFDCA and PHSA. The USDA can regulate under laws like Animal Health Protection Act, Plant Protection Act, Federal Meat, Poultry Products, and Egg Products Inspection Act, and Virus-Serum-Toxin Act. Other agencies that could regulate biotech products are OSHA and CSPC (though they have limited powers) and USFWS and NMFS (through laws like NEPA, ESA, and Lacey Act).

Biotech products are regulated if they have satisfy any of the following conditions a) they contain a plant pest or pesticide, b) they are food, animal or intergeneric microorganism that are used in commerce, or c) they produce a new chemical. However, there are gaps in biotech regulation that need to be addressed. For example, biotech food plants that do not contain a plant pest DNA and are not engineered using a plant pest vector are often not regulated. GE organisms that have only have deletions or single base-pair substitutions (that could be obtained through mutations) or insertions of naturally occurring sequences from sexually compatible relatives (that could be obtained from conventional breeding methods) are also not regulated. These gaps especially exist if a biotech organism falls outside the definition of products that can be regulated by law.
There are also redundancies in regulation. For example, the same GE crop could be reviewed by several agencies for overlapping health, environmental, and agricultural concerns. Different agencies carry out their assessments differently and this could produce different findings that may erode public confidence and confuse developers.
Balancing regulation with innovation
The U.S. regulatory system “protects public health, welfare, safety and environment while promoting economic growth, innovation, competitiveness, and job creation”. Thus there is a balancing of safety through regulations with innovation.
In 1986, the Coordinated Framework for Regulation of Biotechnology called for assessing safety of biotech products while advancing innovation and increasing transparency. Innovation has the potential to enhance safety (for e.g., by replacing high-risk products with newer, safer products) and regulation often fosters innovation (for e.g., fuel economy standards have improved the average fuel economy of vehicles). But if regulation adds substantial upfront costs and delays to the process of developing and marketing a new product, it could impede or delay introduction of new innovative products to the market. This could encourage development of similar (or imitated) products that have already been approved rather than new products that do not have regulatory outlines.
Depending on the product and its intended use, federal oversight of biotech products can happen at a number of phases in the product development cycle. The design-build-test-learn cycle is fundamental for effective and efficient problem solving and is used to develop biotech products (Figure 2). Shortening this cycle can reduce the time it takes to engineer products but it is important to balance this with safety so that both intermediate and final designs can be developed and deployed in a safe manner.

Finally, different statues have different pre-market and post-market requirements and place the burden of proof for safety on different entities (manufacturing company vs regulatory agency). Depending on who is bearing the burden of proof, actions to ensure safety or foster innovation need to be taken.
Risks from biotech products
The committee evaluated five example scenarios to determine if risks from existing biotech products would be similar to risks from future biotech products. For example, can we tackle risks from accidental release of a GE microbial consortium? DIY GE products, an activity which could become more common in the future, are not regulated currently because they are not marketed, sold, or distributed. How would their release in the environment, patterns of use, exposure to children, etc. be regulated? How can we navigate risks from possible off-target effects of genes or transfer of kill switches?
Future products that have similar genetic modifications and use patterns as existing products with a good safety record are likely to have a similar risk profiles. Pilot studies could be carried out to understand risks of releasing GE organisms in the environment (for example, for bioremediation or fuel production purposes). The committee also recommended carrying out probabilistic risk assessments over deterministic as the lack of clarity on the probability of an adverse outcome can lead to disagreements between agencies, the regulated community, and stakeholders on whether or not the assumptions used in a deterministic assessment are sufficient to meet a safety standard and also over the nature and extent of additional data and safety factors needed to address uncertainties.
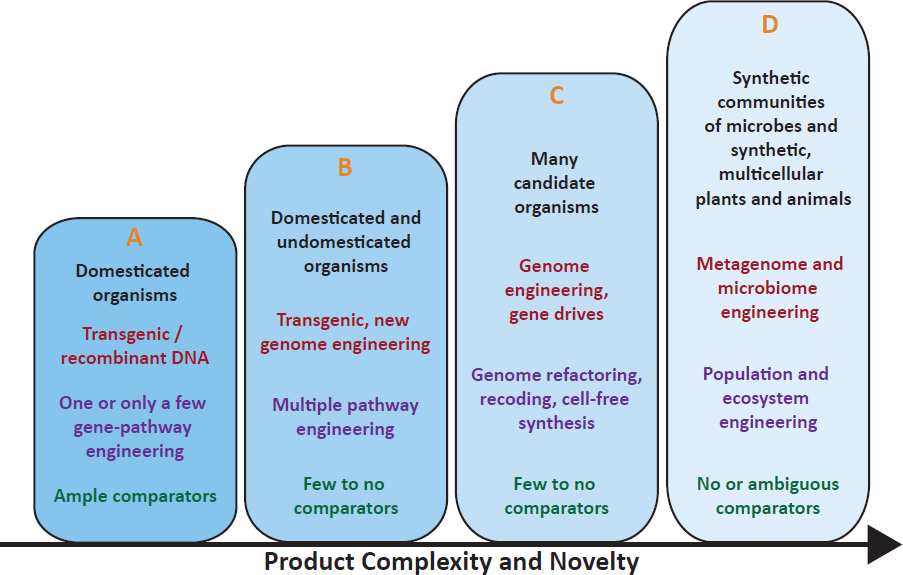
Estimating risks of new products that have no existing products to compare against (comparators) are more challenging. The magnitude of risk may change based on product design and use pattern, and risk pathways could become more complex and multidimensional (Figure 3).
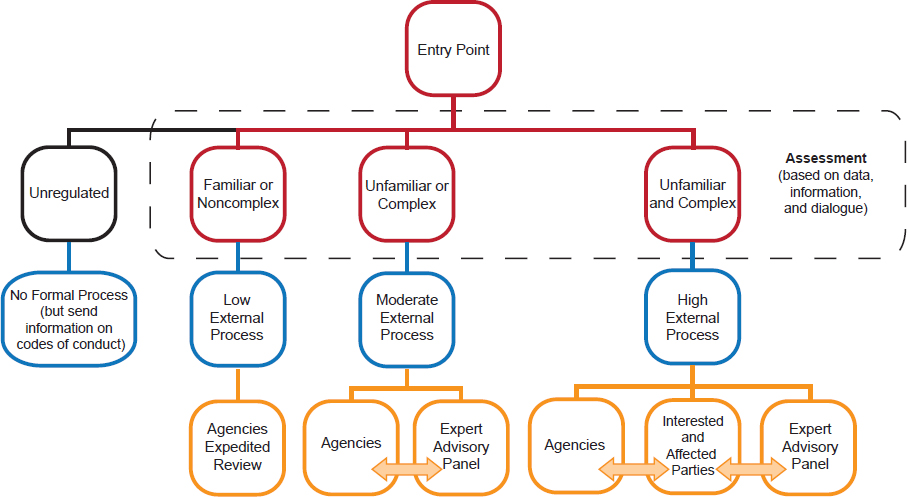
The committee suggested that the familiarity and complexity of new products should determine the regulatory process (Figure 4). The more unfamiliar and complex a new product is, the greater the need for dialogue between different parties, agencies, and experts.
Engaging independent experts and the public
Independent federal advisory committees can assist in many ways including scientific peer review of human health and ecological assessments and providing input on risk assessment methodology and endpoints, environmental modeling, life cycle analyses, sustainability, environmental justice, etc. Agencies like the EPA have been employing external advisory groups and seeking their input and recommendations on several issues for many years now.
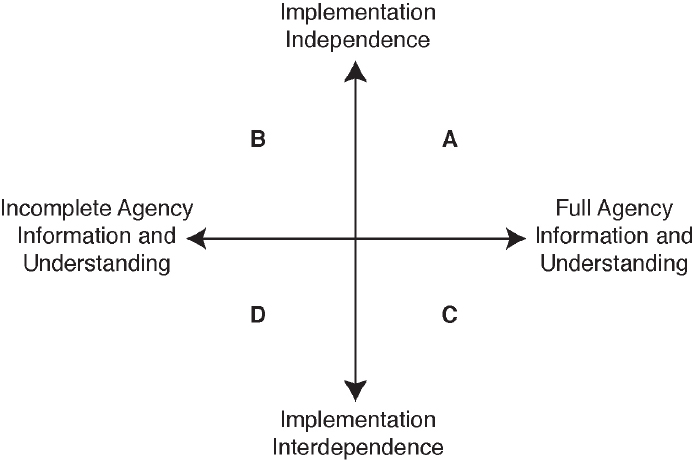
Participation in bureaucracy can vary depending on several factors (Figure 5). Typically, “if an agency has regulatory independence and full knowledge to undertake its regulatory tasks, it could opt to make decisions “in house” and not share knowledge or decision-making logic (quadrant A, “bureaucratic closure”). If the agency has high independence and incomplete knowledge, it could gather information from outsiders “behind closed doors” (quadrant B, “private bureaucratic learning”). In situations where the agency has high interdependence, it could opt to share information, realizing the need to protect confidential business information, and create a process to take in and address additional information (quadrant C, “participatory bureaucratic oversight”). In cases where an agency has high interdependence but limited knowledge on an issue, it could publicly acknowledge its lack of information and initiate a public learning process to inform the future decision (quadrant D, “participatory bureaucratic learning”).”
The committee recommended engaging the public from the start in formulating and characterizing risks for future biotech products. This will increase public confidence in biotech products and provide new perspectives.
Conclusions and main committee recommendations
The bioeconomy is growing rapidly and the increased production of biotech products has the potential to overwhelm U.S. regulatory system in the near future. Regulators will face difficult challenges that go beyond traditional environmental release. The safe use of new biotech products require rigorous, predictable, and transparent risk analyses.
There is a need to advance understanding and use of risk-benefit analyses in biotech products by incorporating external peer review and public participation. A long-term strategy is needed for risk analyses of biotech products and ethical, legal, and social implications of biotech products should be considered. More investments are needed in regulatory science and in graduate and post-graduate knowledge of the regulatory system.

