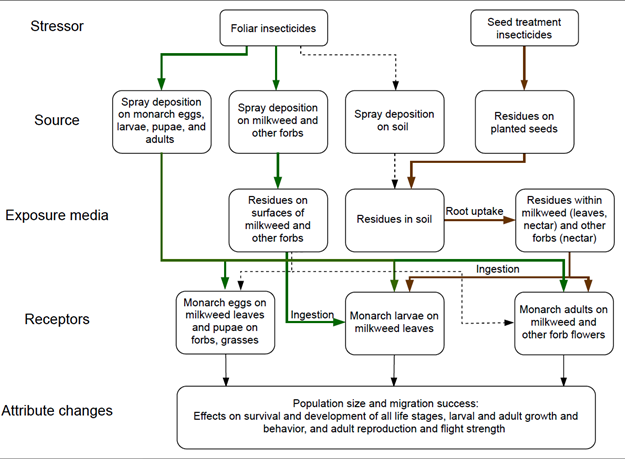
Featured image: A conceptual model to explain how monarch butterflies could be exposed to insecticides used in Midwest agriculture. Figure obtained from Krishnan et al. (2021).
As an insect toxicologist, I am excited to write on this subject! Since a lot of insect toxicology work, especially on pollinators and other non-target species, are conducted by non-toxicologists, I hope this post will be useful to the many non-experts working in the field.
While this post focuses on insects and laboratory bioassays, many components also apply to other kinds of ecotoxicology studies. And several of the guidelines apply to all experimental studies.
- Determine the goal of the study prior to designing the experiment. This is an obvious one to experimental scientists. If your goal is to study how a pesticide impacts a species, you might want to study a number of endpoints (e.g., mortality, growth, development, reproduction, behavior, etc.). If, however, your goal is to identify which of several pesticides is most toxic to the species, you might study just one or two endpoints (typically mortality and growth) that can help rank toxicity.
- Gather as much background information on the chemical and organism to be tested. Answer these questions before you start your experiments: Why this particular chemical (does the test organism have high likelihoods of exposure, is it a part of your research question)? Where, how, and in what quantities is the chemical used? What are the chemical’s properties (solubility, volatility, bioaccumulation potential, half-life, etc.)? Is it highly toxic to humans (if so, handle with care)? Your choice of organism is also dependent on your question. A pest organism is used to study a pesticide’s efficacy while a non-target organism is used to study its negative impacts. For obvious reasons, easy-to-rear and abundantly found organisms are preferred. If you are generating data for regulatory needs, the regulators might have provided a list of acceptable test organisms. A model organism like Drosophila can be ideal if you plan to manipulate the organism at the molecular/cellular level. Regardless of the organism chosen, it is important to understand its life cycle, developmental stages, natural habitat, needs (food, environmental conditions), behavior, rearing techniques, etc. to successfully conduct experiments with it.
- Generate conceptual models when possible. By ensuring you consider possible sources, routes, and stages of exposure, conceptual models can help design studies. The feature image shows the conceptual model I generated to explain how monarch butterflies can be exposed to insecticides used in Midwestern agricultural fields.
- Consult with a statistician. Many people mistakenly believe that consultation with statisticians should only occur after the data have been collected. Statisticians are also very helpful in ensuring your experimental design will help answer your question. They will discuss importance of randomization (ideally, planned randomization at every step of the experiment), difference between experimental and observational units, minimum number of replicates needed (number of insects needed per replicate and number of replicates needed per experiment), types of data analyses possible for the experimental set-up, etc. Of course, definitely consult a statistician (preferably the same) before analyzing the collected data too. In my experience, information from basic statistics classes often cannot be directly applied as real-world data is messy and complex. Plus, statistical tests can change with the research question and experimental set-up.
- Determine the doses or concentrations to be tested. There are three ways you can choose your doses/concentrations. If you would like to generate a dose (or concentration)-response curve, choose approximately 5 doses/concentrations that are 3X to 10X apart and that cover a range of effects – for example, from no mortality to 100% mortality. Another option is to select concentrations based on the organism’s potential exposure concentrations – these can be mean, median, maximum, 10X, or 100X maximum concentrations of the compound found in the field. Finally, the highest dose that doesn’t cause an adverse effect or the lowest dose that causes an adverse effect can be chosen. This, however, is done typically for chronic mammalian studies where regulatory frameworks require identification of safe doses. Generally, for insect toxicology studies, it is beneficial to first generate dose-response curves.
- Determine appropriate units and matrices. Unfortunately, in insect dietary bioassays, solution concentrations are often used as surrogates for leaf/diet concentrations. This equivalency is flawed; the sweetness of a pie glaze is not merely determined by the concentration of the sugar solution. It is also determined by the volume of sugar solution and the surface area of the pie to which it is applied. Avoid using terms like ppm (parts per million) and ppb (parts per billion) as these units can correspond to any matrices, i.e., weight/weight, weight/volume, volume/weight, volume/volume, etc. Thus, be explicit about units (ng/g) and matrices (ng of insecticide/g leaf). Finally, ‘dose’ and ‘concentration’ should not be used interchangeably.
- Choose realistic exposure duration, observation period, and toxicity endpoints. Consider physio-chemical properties and half-lives of the chemical, duration and behavior of insect stage being treated, and potential for chemical exposure. Gather as much data/observations as possible but have mechanistic rationale for difficult-to-collect endpoints. While it is perfectly plausible that you might discover a novel chemical effect, collecting endpoints that require a lot of investment (time, resources, and money) in the absence of a mechanistic rationale might prove more disappointing than fulfilling.
- Choose the right solvent(s). Choosing the right solvent is critical in a toxicity bioassay. Obviously the solvent should be non-toxic to the organism. Additionally, complete solubility of the chemical in the solvent at room temperature will likely ensure a uniform solution concentration. The route of chemical exposure is another important aspect; for topical bioassays in insects, acetone is more appropriate than water as water cannot penetrate the lipids in the cuticle. For dietary bioassays, where the chemical is applied on a leaf, a surfactant might be necessary to ensure complete spread of the chemical on the leaf’s surface.
- Choose appropriate control(s) and account for control mortality. While the inclusion of positive controls in bioassays is highly desirable (it can inform if an insect batch is showing an atypical response), the absence of a negative control often invalidates the experiment. A negative control can be two kinds: untreated insects and/or insects treated only with the solvent. The latter is recommended as it will inform solvent effects and whether the solvent has been inadvertently contaminated (which means your chemical treatments are also likely contaminated). If a formulated product is being used to understand the effects of a pesticide active ingredient, the negative control must contain the formulated product minus the active ingredient. In mixture studies where multiple chemicals are combined, it is important to conduct bioassays with individual chemicals to understand which chemical(s) contributes to the insect’s response to the mixture. Ideally, control mortality must not significantly exceed the background mortality and must be accounted (if both the control and lowest dose cause 20% mortality, the lowest dose is assumed to cause no toxic effect). Formulas like Abbott’s can help correct for control mortality.
- Use high purity chemicals and healthy insects. Chemicals should have high purity and be stored under the right conditions (while colder is not always better, darker is almost always better). Check the expiry date periodically and keep the safety sheets handy. Ideally, insects should be used from well-established colonies that are healthy and free from chemical exposures, parasites/infections, genetic abnormalities, stress, and lengthy periods of inbreeding. First-generation insects should be avoided as they could have had prior exposure to stressors. Homogenous larvae (similar stage/weight) are more likely to provide homogenous responses.
- Standardize experimental conditions. Insects should be reared under optimal and consistent temperature, humidity, and light settings. Ad libitum food should be provided. If the food being provided is plants, ensure the plants are healthy and are reared similarly throughout the experiment. If the food being provided is an artificial diet, ensure it is unexpired and prepared and stored the same way throughout the experiment.
- Decide what data can be excluded prior to start of experiment. Prior to initiating definitive bioassays, it is important to know how the species or colony behaves in the lab. What is the typical background mortality? How many days does it take to develop? What constitutes as normal behavior? How much food does it need? Certainly there will be variations across generations but understanding the range of responses can help define the norm for the species or colony, and can be used as a “cutoff” for an acceptable bioassay. Thus, if the control response deviates from the norm – for example, the control mortality exceeds the high-end background mortality rate of 30% or the controls take twice as long to develop or they feed significantly less – the bioassay run can, and should, be justifiably excluded. A bioassay should not be excluded due to variable responses in the treatment group.
- Note qualitative observations. Scientists prefer quantitative data for obvious reasons. However, qualitative data also have value; the lethargic or paralytic response of an organism informs us that the chemical is causing an effect, whether or not it results in mortality or sublethal effects. And while the organism might eventually recover by metabolizing and excreting the chemical, the time spent ‘defenseless’ has real-world consequences; in the wild, the organism might have more easily been parasitized or predated upon or might lose out on food or mate. Also, if the symptoms don’t match up with the chemical’s known mode of action and expected responses, you might have found a novel toxicity pathway (like I recently did)!
- Verify concentrations in different matrices. It is crucial to verify the concentrations of chemical matrices for two reasons: you might have prepared an incorrect concentration (errors in weighing scales, pipettes, calculations, etc.) or you might have contamination (use of non-disposable or inadequately clean supplies, contamination of reagents, chemical, etc.). Often, it is better to analyze a matrix (like a chemical solution) before the start of a bioassay – otherwise, the presence of a contaminant would necessitate rerunning of the experiment. And it is best to analyze as many matrices (solution, leaves, diet, insects, etc.) and samples (replicates across multiple concentrations and time periods) as possible; however, this can be cost-prohibitive. Regardless, it is important to analyze at least three samples in a matrix to ensure the test organisms were provided the right (within a reasonable margin of error) concentration/dose.
- Provide detailed methods and analyses and make raw data and metadata publicly available. Science faces a reproducibility crisis; in certain fields, less than half of published results can be replicated. Providing detailed information on methods and analyses can allow other scientists to replicate your findings or identify flaws. Making available the raw data and metadata can help other scientists to better understand your data, reprocess it for accuracy, identify errors, or use it for other purposes (you will get cited!). It can also boost confidence in your research. While detailed methodological or analyses information should be provided in the main paper or supplementary files, raw data are often uploaded onto publicly accessible repositories like those hosted by the journal, universities, or open-source sites like GitHub.