Featured image: A ripple effect on water. Image source: Sergiu Bacioiu from Romania [CC BY 2.0 (https://creativecommons.org/licenses/by/2.0)]
Think about the ripple effect. Where one thing causes a series of other things to happen, and the small changes lead to a big impact.
Toxicity Pathways (TPs) and Adverse Outcome Pathways (AOPs) are two approaches in toxicology which accommodate this ripple effect. They represent small biological changes leading to bigger and more harmful effects.
Toxicity Pathways
TP is a sequence of events, starting with a chemical entering a person. A proportion of this chemical reaches the target tissue and interacts with molecules in it — for example, by binding to cell receptors (similar to a key finding the right lock) . This causes a perturbation, or disturbance, in the normal functioning of the cell. The cell starts to alter, and if the body corrects the changes in time, there will be no problems. But if the body doesn’t, the cell will be permanently altered/injured. This could lead to the exposed person being harmed. Figure 1 represents this diagrammatically.

In the Toxicity Testing for the 21st Century report, the scientists believed that evaluating TPs for biologically significant disturbances could become the new system for testing toxicity. Generally speaking, if is a cell is injured by a chemical, computer models can be used to predict if this will affect a whole organism. This would greatly reduce animal testing.
Adverse Outcome Pathways
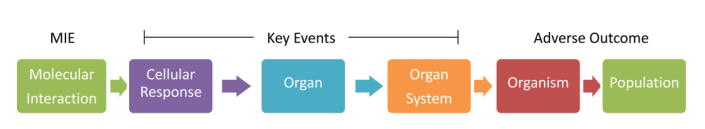
AOPs encompass TPs and extend further. That is, they include chemical-molecular interaction and cellular changes, but also look at how this leads to changes in organs, organisms and populations. It’s a true ripple effect. Population-level AOPs are generally used for non-human species, where the goal is not to protect every organism but to protect the species (or a population) as a whole.
Figure 2 shows what altered responses in each stage could look like. For example, at the cellular stage, an alteration could be the production of abnormal proteins. These proteins could cause the organ to not function properly. If the organ affected is a testicle, then fertility could be impaired. And if most males in a population become infertile, the sex ratio would become skewed.

Figure 2 also indicates a molecular initiating event (MIE), which are molecular interactions. MIEs, along with key events (KEs), which are cellular and organ responses, are important in AOPs (Figure 3). If two different chemicals have the same MIE and KE, they will likely produce the same adverse outcome (AO) on an organism and its population. Thus, both chemicals need not be tested on whole organisms, minimizing animal use.
Conclusions
Both TPs and AOPs were developed for similar reasons, i.e., to understand how a chemical’s interaction with biological molecules could lead to harmful effects. Nowadays even reverse AOPs (AOs to MIEs) are being constructed when sufficient molecular interaction data are not available. Also, there are currently initiatives to link distinct AOPs through their MIEs, KEs and AOs.
Generally, TPs are more detailed than AOPs. Both can be quantitative, meaning separate pathways can be constructed for different doses of a chemical (for example, some chemicals can cause different adverse outcomes at low and high doses).
Both TPs and AOPs are knowledge-driven, employ computational technologies, and strive to capture the big picture. These make them ideal tools for toxicology in the 21st century.


1 Comment